Thermoplastic polymers are reshaping wearable health devices by addressing critical challenges like skin irritation, material degradation, and long-term safety. These materials, particularly thermoplastic polyurethanes (TPUs), offer flexibility, durability, and breathability, ensuring comfort and reliability for continuous use. Unlike traditional rigid materials, thermoplastics can flex with the body, resist sweat-induced breakdown, and maintain biocompatibility standards like ISO 10993.
Key takeaways:
- Skin-Friendly: Thermoplastics prevent irritation and allergic reactions by avoiding harmful solvents and plasticizers.
- Durable: Resistant to sweat, body fluids, and UV exposure, ensuring longevity.
- Comfortable: Flexible and breathable, reducing discomfort during extended wear.
- Eco-Conscious Options: Emerging biodegradable thermoplastics like genTPU reduce environmental impact.
AIH LLC incorporates these materials into devices like the aiSpine posture monitor and aiRing vital signs ring, ensuring user safety, comfort, and precise monitoring. With advancements in material science, thermoplastic polymers continue to drive the evolution of wearable health technology.
Biocompatibility Challenges in Wearable Health Devices
Skin Irritation and Allergic Reactions
One major hurdle for wearable health devices is preventing skin irritation and allergic reactions. For instance, acrylic adhesives – commonly used in these devices – can lead to contact dermatitis. Additionally, leftover monomers, plasticizers, and by-products from polymers might cause local cytotoxicity or even systemic toxicity. Materials that fail to adapt to the skin’s natural movements can also create problems. Stiff or abrasive components may lead to pressure sores and microtrauma due to constant friction. Another issue is galvanic corrosion, where sweat acts as an electrolyte, causing chemical reactions that result in skin rashes and, in some cases, device malfunction.
Material Degradation from Sweat and Body Fluids
Sweat and body fluids present a tough challenge for the materials used in wearables. Over time, prolonged exposure to these fluids can lead to hydrolysis and chemical degradation, which weakens materials and compromises both their safety and performance. Devices with occlusive patches trap humidity levels as high as 80%–95%, accelerating material breakdown. This trapped moisture can soften the skin, a condition known as maceration, which increases friction and creates an ideal environment for bacteria. Even commensal bacteria like Staphylococcus spp. thrive at relative humidity levels as low as 81%–87%, which are easily reached under non-breathable devices. According to Timothy Frisch and Justin Spitzer from Covestro, materials need to resist hydrolysis caused by sweat, body fluids, and moisture to maintain their integrity. These challenges underscore the importance of developing better materials for wearables.
Long-Term Safety and Cytotoxicity Concerns
The risks don’t end with short-term irritation – long-term use introduces additional safety concerns. Extended wear can lead to issues like sensitization, fibrotic encapsulation, or foreign-body responses. Michael Swartz, Director of Business Development at Mighty Studios, highlights the importance of ongoing validation:
"Compliance is not a one-time checkbox; it requires careful upfront material selection and iterative validation throughout product development".
For clinical devices, the stakes are even higher. They must meet stringent leakage-current standards, such as those outlined in IEC 60601-1, to ensure safety during activities like sweating or repetitive motion. Without rigorous material testing, devices might release harmful irritants or fail electrically after weeks or months of continuous use. Addressing these concerns is key to advancing thermoplastic polymer solutions for wearable health devices.
sbb-itb-44aa802
Selecting TPU For Medical Device Applications
How Thermoplastic Polymers Solve Biocompatibility Problems
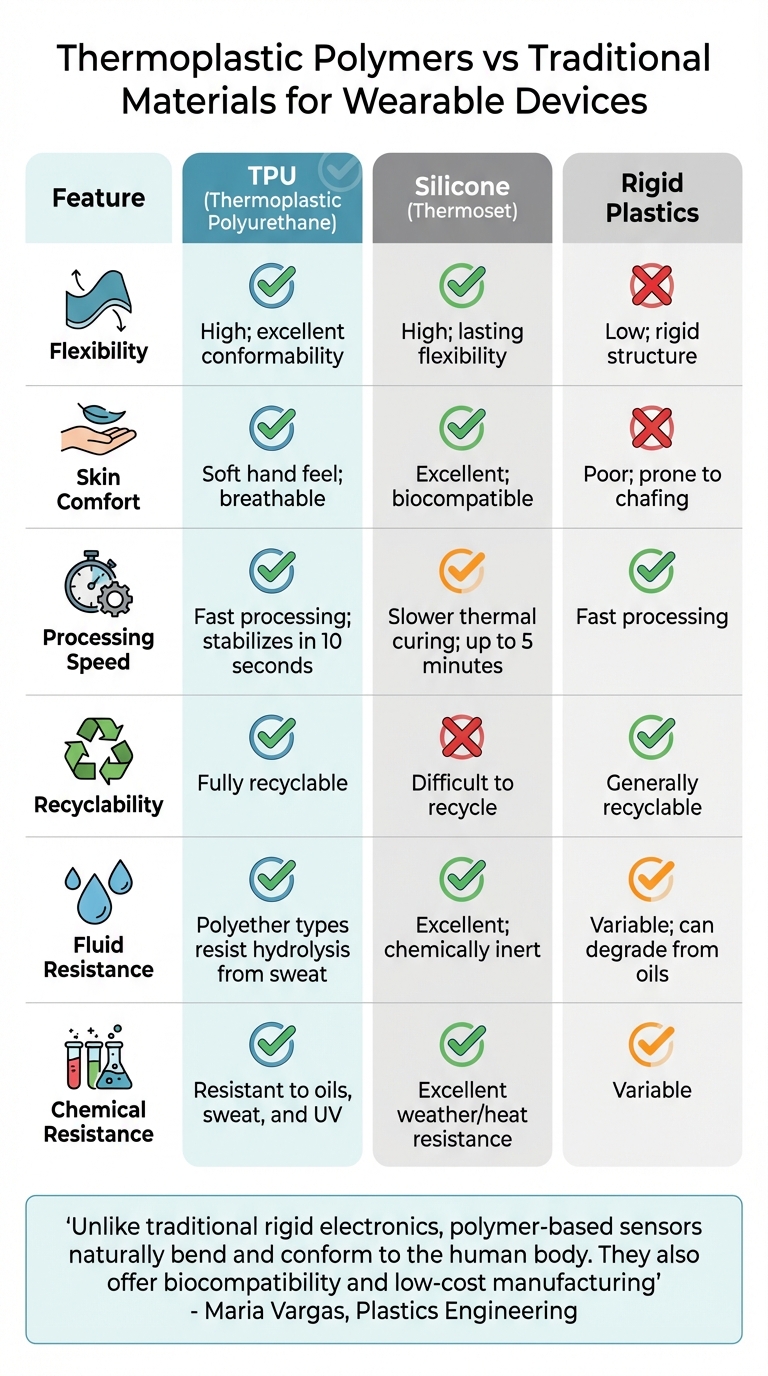
Thermoplastic Polymers vs Traditional Materials for Wearable Devices
Properties of Thermoplastic Polymers
Thermoplastic polymers bring a range of qualities that tackle biocompatibility concerns head-on. Unlike other materials, these polymers can be made without solvents or plasticizers, which are common culprits behind chemical leaching and allergic reactions. One standout feature is their breathability – TPU films allow water vapor to pass through while keeping liquids out. This prevents moisture buildup, reducing irritation and discomfort. Additionally, their flexibility and breathability help minimize friction, while polyether-based TPU films are designed to withstand hydrolysis and UV exposure, ensuring they remain safe for long-term skin contact.
Thermoplastic Materials Used in Wearables
Thermoplastic polyurethane (TPU) formulations are particularly effective at addressing challenges like skin irritation and material degradation in wearable devices. The wearable health device market relies on medical-grade thermoplastics for their unique properties. For example, Desmopan® and Platilon®, TPU films developed by Covestro, are well-established in applications like adhesive mounting, top cover layers, and electronic substrates. Some advanced TPUs, like DiAPLEX MM3510, feature a glass transition temperature of 95°F (35°C). This allows them to adapt their shape near body temperature, offering a better fit and enhanced comfort. The growing demand for polymer-based sensors in healthcare flexible electronics is projected to drive this sector to $9.3 billion by 2025. To better understand the advantages of these materials, here’s a comparison with traditional options.
Thermoplastic Polymers vs. Traditional Materials
Thermoplastic polymers demonstrate clear advantages over traditional materials, especially for wearable applications. Here’s how they stack up:
| Feature | Thermoplastic Polymers (TPU) | Silicone (Thermoset) | Rigid Plastics |
|---|---|---|---|
| Flexibility | High; excellent conformability | High; lasting flexibility | Low; rigid structure |
| Skin Comfort | Soft hand feel; breathable | Excellent; biocompatible | Poor; prone to chafing |
| Processing Speed | Fast processing; stabilizes in 10 seconds | Slower thermal curing; up to 5 minutes | Fast processing |
| Recyclability | Fully recyclable | Difficult to recycle | Generally recyclable |
| Fluid Resistance | Polyether types resist hydrolysis from sweat | Excellent; chemically inert | Variable; can degrade from oils |
| Chemical Resistance | Resistant to oils, sweat, and UV | Excellent weather/heat resistance | Variable |
Maria Vargas from Plastics Engineering highlights these advantages, saying:
"Unlike traditional rigid electronics, polymer-based sensors naturally bend and conform to the human body. They also offer biocompatibility and low-cost manufacturing".
With their flexibility, skin-friendly properties, and durability, thermoplastic polymers are setting the standard for the next generation of wearable health devices.
Thermoplastic Polymers in AIH LLC Wearable Devices
AIH LLC has embraced thermoplastic polymers in its wearable devices to improve both safety and comfort for users. These materials offer an ideal combination of biocompatibility, flexibility, and durability, making them a perfect fit for health monitoring technology.
aiSpine Posture Monitoring Device

The aiSpine posture monitoring device incorporates medical-grade thermoplastic elastomers to deliver all-day comfort and safety. These elastomers adhere to ISO 10993-10 (irritation-free) and ISO 10993-5 (cytotoxicity) standards, ensuring they are gentle on the skin during extended wear. The device’s thermoplastic housing features a soft, velvety surface that eliminates the irritation often caused by rigid plastics. This design makes it suitable for various wearing styles, whether clipped to clothing or placed directly on the skin.
Additionally, advanced thermoplastic copolyesters used in the aiSpine provide excellent durability and shape retention. These materials adapt seamlessly to the spine’s contours and resist wear, such as material fatigue or delamination, even with repeated use and contact with sealing tapes. The housing is also designed to be breathable, reducing sweat buildup during prolonged wear and enhancing user comfort.
aiRing Vital Signs Monitoring Ring
For the aiRing vital signs monitoring ring, AIH LLC uses thermoplastic polyurethane (TPU), a highly durable and flexible material. TPU ensures the ring can handle continuous skin contact without compromising its performance. Its stretchable properties allow the ring to fit comfortably on various finger sizes while maintaining the precision needed for accurate sensor readings.
The aiRing also features shape-memory polymers that respond to body temperature (98.6°F/37°C), enabling the ring to self-adjust for a snug, custom fit. This ensures reliable contact between sensors and the skin, which is crucial for accurate vital signs monitoring. Additionally, the thermoplastic housing is resistant to chemicals like hand sanitizers and soaps, ensuring the ring’s longevity even with frequent exposure to everyday substances. These innovations not only enhance durability but also make the aiRing a reliable tool for health monitoring.
Upcoming AIH Device Applications
Building on these advancements, AIH LLC is developing new devices like aiNeuro, designed for stroke monitoring and posture correction. This upcoming device will utilize cutting-edge thermoplastic formulations that combine soft-touch surfaces for comfort with the mechanical strength required for continuous monitoring. These materials will be particularly suited for neck and head applications, ensuring optimal biocompatibility and performance.
The use of conductive polymer composites (CPC) in future devices will further expand capabilities by enabling high stretchability for motion monitoring without compromising material integrity. Such advancements will allow for seamless sensor integration while maintaining the skin-friendly properties that define AIH’s wearable devices. The global biomaterials market, which is expected to grow to $88.4 billion at a 15% compound annual growth rate, underscores the potential for these innovations. AIH’s focus on thermoplastic technology ensures its products remain at the forefront of wearable health monitoring, meeting rigorous FDA standards for medical-grade devices.
Future Developments in Thermoplastic Polymers for Medical Wearables
Eco-Friendly Thermoplastic Formulations
Sustainable materials are reshaping the future of wearable health devices. In December 2025, Ecogenesis Biopolymers introduced genTPU, a thermoplastic polyurethane filament derived from plants. This material reduces its carbon footprint by an impressive 83.8% compared to petroleum-based options. Testing under modified ASTM D5338 standards revealed 27% biodegradation within 180 days at 113°F (45°C). This demonstrates that eco-conscious thermoplastics can maintain the durability and flexibility required for medical applications while significantly lessening their environmental impact.
"genTPU offers the mechanical strength and flexibility expected from conventional polyurethane while delivering a significantly lower environmental footprint."
– Ecogenesis Biopolymers
In addition to genTPU, natural biopolymers like chitosan and cellulose are gaining attention as renewable options. However, these materials often need bio-based plasticizers, such as sorbitol or glycerol, to achieve the elasticity required for skin-conforming devices. Meanwhile, polyhydroxyalkanoates (PHAs), sourced from microbial processes, address concerns about marine plastic pollution due to their ability to biodegrade in ocean environments. With global e-waste reaching 62 million tons in 2022 – an 82% jump since 2010 – and only 22% of it being formally recycled, the demand for biodegradable "transient" electronics is growing rapidly. These advancements in materials are crucial for meeting regulatory demands while addressing environmental concerns.
Meeting FDA and Regulatory Standards
Developing biocompatible materials for wearables also requires navigating strict regulatory landscapes. The FDA and similar organizations mandate extensive biocompatibility testing, evaluating factors like toxicity, allergic reactions, and immune responses, before approving wearable devices for market use. Polymers such as polylactic acid (PLA), polycaprolactone (PCL), and poly(lactic-co-glycolic acid) (PLGA) offer adaptable mechanical properties and degradation rates, making them suitable for meeting these stringent safety and performance requirements.
The rise of 3D-printed components introduces additional hurdles, as manufacturers must ensure that residual photoinitiators and unreacted monomers do not compromise biocompatibility. Thermoplastic shape memory polymers provide a promising solution, as their linear molecular structures allow for reprocessing and recycling, addressing both sustainability and safety concerns. Moreover, Life Cycle Assessment (LCA) frameworks, outlined under ISO-14040 and ISO-14044 standards, are gaining importance. These frameworks ensure that the entire lifecycle of medical polymers, from production to disposal, adheres to environmental and safety benchmarks.
Combining Thermoplastic Polymers with AI Monitoring
Advances in thermoplastic formulations are now being paired with AI-driven sensor technologies, leading to smarter, more efficient wearables. For instance, in December 2025, researchers at the University of Rome "Tor Vergata" developed a hybrid TPU composite by combining multi-walled carbon nanotubes (MWCNTs) and aluminum nanoparticles (AlNPs) in a 5:1 ratio. This blend achieved a balance of stiffness and thermal responsiveness, enabling effective shape-memory actuation. Similarly, fluorine-rich supramolecular polyurethanes have been designed to stabilize ion transport, allowing sensors to maintain strong mechanical properties – approximately 32.31 MPa in strength and 107.05 MJ/m³ in toughness – even under extreme conditions as low as –40°F (–40°C). These materials also achieved a high temperature coefficient of resistance (TCR) of 8.05% °C⁻¹ under cryogenic conditions.
These breakthroughs offer companies like AIH LLC the opportunity to elevate their wearable health devices. By integrating biocompatible thermoplastic polymers with advanced sensor technologies, they can deliver devices that are both high-performing and safe for long-term use. This approach highlights AIH LLC’s dedication to combining cutting-edge materials with real-time monitoring to enhance wearable functionality and reliability.
Conclusion
Key Benefits of Thermoplastic Polymers
Thermoplastic polymers play a critical role in improving the performance and comfort of wearable devices. Materials like TPU and TPE address biocompatibility challenges by eliminating solvents and plasticizers that can irritate the skin. Their breathability allows air and moisture to circulate, helping to prevent issues like skin maceration. Additionally, these polymers are up to 10 times lighter than traditional metals and provide excellent resistance to sweat, body fluids, and environmental stressors.
Their flexibility allows wearable devices to adapt to complex body contours, ensuring consistent sensor contact. At the same time, their resistance to tearing and chemicals enhances durability. Advanced manufacturing methods further enable the creation of ergonomic designs that prioritize user comfort.
"TPU films represent a valuable enhancement for wearable medical devices. Their unique combination of flexibility, stretchability, biocompatibility and manufacturability makes them an optimal choice for the next generation of printed electronics in healthcare." – Timothy Frisch and Justin Spitzer, Covestro
These features collectively contribute to the exceptional performance and reliability of wearable devices.
AIH LLC’s Use of Advanced Materials
AIH LLC leverages the benefits of advanced thermoplastics in its wearable technologies. The aiSpine posture monitoring device and aiRing vital signs monitoring ring utilize these skin-friendly, durable materials to deliver precise sensor performance and all-day comfort for users.
FAQs
How can I tell if a wearable material is truly biocompatible?
To ensure a wearable material is safe for use on the body, it must pass rigorous biocompatibility testing. This process checks how the material interacts with the body, focusing on factors like irritation, sensitization, and toxicity. It also requires compliance with regulatory standards such as ISO 10993-1.
Crucial tests include cytotoxicity, which evaluates whether the material is harmful to cells, and hemocompatibility, which ensures it won’t negatively affect blood. Additionally, chemical properties are analyzed to prevent issues like harmful substance leaching or bacterial growth. These steps are essential to confirm the material is suitable for long-term skin contact.
Why is TPU better at handling sweat and daily wear over time?
TPU stands out for its ability to endure sweat and daily wear, thanks to its flexibility, stretchability, and resistance to abrasion, chemicals, and oils. These features make it both durable and comfortable, even during extended use.
Are biodegradable thermoplastics safe for long-term skin contact?
Biodegradable thermoplastics, when carefully formulated, can be suitable for long-term skin contact. These materials are crafted to be both biocompatible and flexible. However, their safety largely depends on factors like their composition and the specific way they’re intended to be used. It’s crucial to ensure the material complies with all relevant biocompatibility standards for your particular application.