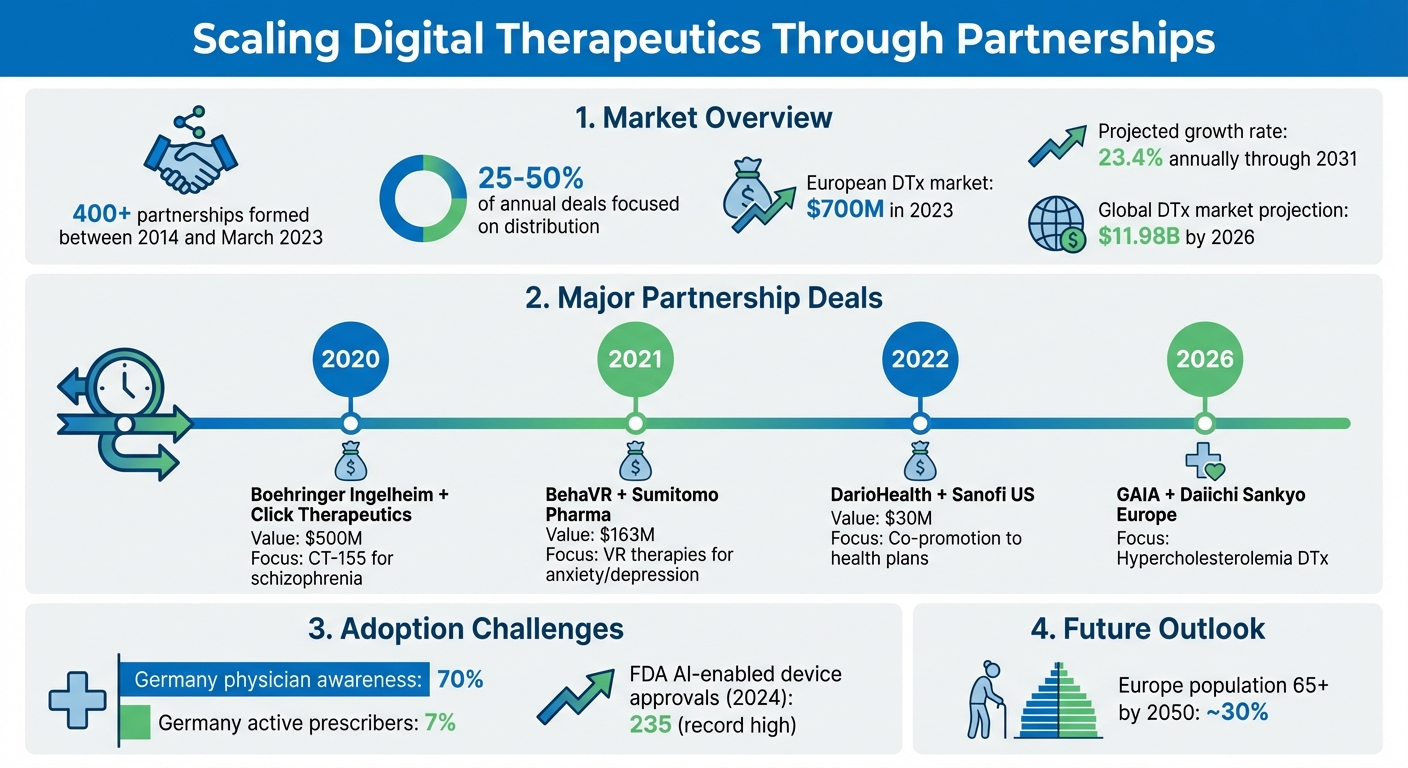
Scaling digital therapeutics (DTx) globally requires collaboration. Between 2014 and March 2023, over 400 partnerships were formed, addressing challenges like regulation, reimbursement, and adoption. These alliances – ranging from pharmaceutical companies to health systems – help bridge the gap between innovative digital solutions and patient impact.
Key Takeaways:
- What Are DTx? Software-based, clinically validated tools for managing or treating medical conditions, often prescribed by healthcare providers.
- Why Partnerships Matter: They provide resources, expertise, and networks for scaling, with 25–50% of deals focused on distribution.
- Success Stories: Examples include DarioHealth and Sanofi ($30M deal in 2022) and BehaVR with Sumitomo Pharma ($163M deal in 2021).
- Regulatory Challenges: Partnerships help navigate approval and reimbursement, as seen with GAIA and Daiichi Sankyo‘s collaboration in 2026.
Next Steps for DTx:
- Tailor solutions for diverse healthcare systems.
- Prioritize data privacy and security to build trust.
- Strengthen long-term collaborations with providers and regulators.
The future of DTx depends on partnerships that balance technical innovation with practical healthcare needs, ensuring solutions reach patients effectively.
Digital Therapeutics Partnerships: Key Statistics and Success Stories 2014-2026
579 – Scaling Predictive Healthcare: How No Code Platforms Drive Personalised Digital Therapeutics
sbb-itb-44aa802
Lessons from Successful Digital Therapeutics Partnerships
Building on earlier insights, let’s dive into some key takeaways from successful digital therapeutics (DTx) partnerships.
Aligning on Shared Goals and Values
The best DTx partnerships begin with clear, shared objectives. This means ongoing discussions about evidence generation, target patient populations, and success metrics. A great example is the March 2022 partnership between DarioHealth and Sanofi US. This multi-year, $30 million agreement combined Dario’s agile software development with Sanofi’s expertise in evidence generation and market research. Together, they co-promoted solutions to health plans, aligning their strategies from the start to meet both regulatory and commercial needs.
This collaboration highlights the importance of bridging operational differences. Pharmaceutical companies often follow long-term, structured processes, while DTx firms thrive on rapid, iterative cycles. Successful partnerships respect these differences and find ways to integrate both approaches effectively.
Combining Complementary Strengths
DTx partnerships thrive on combining expertise that neither party could achieve independently. Pharmaceutical companies bring clinical knowledge, regulatory experience, and resources for large-scale trials. On the other hand, DTx firms excel in areas like AI, virtual reality, and real-time data analytics.
Take the December 2021 agreement between BehaVR and Sumitomo Pharma, valued at up to $163 million. This partnership focused on developing virtual reality therapies for social anxiety and major depressive disorder. Sumitomo contributed $5 million upfront and additional R&D support, while BehaVR provided the technical VR platform.
"We had the subject matter expertise in those clinical areas, and BehaVR clearly had the technical expertise. So, it’s a very synergistic partnership that makes sense to commit to for the long term." – Krys Zaluski, Director of Business Development, Digital Health, Sumitomo Pharma
Another notable example is the 2020 collaboration between Boehringer Ingelheim and Click Therapeutics. This $500 million deal aimed to develop CT-155, a digital therapeutic for schizophrenia. Click contributed its proprietary platform technology, while Boehringer brought its neuropsychiatric clinical expertise and commercialization capabilities. These partnerships show how complementary strengths fuel innovation, though navigating regulatory complexities remains a shared challenge.
Addressing Regulatory and Reimbursement Challenges
Regulatory approval and reimbursement are two of the toughest hurdles for DTx companies. Partnerships can help by combining resources and expertise to tackle these systematically. For instance, in March 2026, GAIA and Daiichi Sankyo Europe teamed up to commercialize "lipodia", a DTx for hypercholesterolemia. GAIA led the Phase 3 trial and regulatory submission under Germany’s DiGA pathway, while Daiichi Sankyo contributed cardiovascular expertise and supported market expansion across Europe.
"Through our partnership with Daiichi Sankyo Europe, we are in the best position to integrate this evidence-based digital therapy into routine care in the near future, achieving the reach, scientific credibility, and impact that patients and physicians expect." – Dr. Mario Weiss, CEO, GAIA
Securing reimbursement is another critical focus. Partnerships often work to integrate DTx solutions into existing healthcare systems, transitioning them from consumer apps to prescription-based models. This shift is essential for long-term adoption and sustainability.
Strategies for Scaling Digital Therapeutics Globally
Adapting Solutions for Different Healthcare Markets
Expanding digital therapeutics (DTx) across international markets means tailoring solutions to fit each region’s unique healthcare systems, regulations, and reimbursement structures. For instance, the European DTx market was valued at around $700 million in 2023 and is expected to grow at an annual rate of 23.4% through 2031. Tapping into this growth requires careful navigation of diverse regional requirements.
One strategy is to design products that comply with the most stringent regulatory standards, such as Japan’s PMDA or the EU’s MDR. Meeting these benchmarks early can simplify entry into multiple markets. Alternatively, focusing on markets with more flexible regulatory frameworks, like the US FDA or UK MHRA, can speed up the process. These agencies offer tools like regulatory sandboxes and Predetermined Change Control Plans (PCCP), which allow for quicker iterations.
Another effective approach is implementing tiered evidence systems. For example, real-world evidence (RWE) or observational studies can support conditional approvals or justify lower reimbursement rates. Meanwhile, randomized controlled trials (RCTs) are often necessary for securing higher reimbursement levels. A case in point is the UK’s National Institute for Health and Care Excellence (NICE), which approved Sleepio, an insomnia app, in May 2022 based on its economic and cost-effectiveness benefits rather than solely on clinical outcomes.
Building Trust Through Data Privacy and Security
When scaling DTx globally, ensuring robust data privacy and security is non-negotiable. Trust from patients, healthcare providers, and regulators depends on transparent data practices and strong privacy measures. Without these, stakeholder confidence can erode.
Compliance with region-specific regulations is essential. For example, in the United States, companies must adhere to HIPAA standards, while in Europe, GDPR compliance is mandatory. Additionally, the EU AI Act classifies healthcare AI systems as high-risk technologies, requiring thorough assessments and transparency. In Singapore, the Cybersecurity Labelling Scheme provides a four-tier framework to standardize cybersecurity requirements for digital health tools.
Incorporating cybersecurity measures during product development can streamline market entry and enhance a product’s reputation. In 2024, the Digital Therapeutics Alliance (DTA) collaborated with DirectTrust to establish accreditation programs specifically for verifying data privacy in DTx platforms. Advanced encryption methods and access control protocols, like those outlined by HL7 FHIR standards, are crucial for safeguarding patient data and maintaining confidentiality. With these measures in place, companies can foster trust and enable long-term partnerships.
Establishing Long-Term Collaboration
Sustained growth for DTx depends on fostering strong, ongoing relationships with healthcare providers and regulators. Healthcare professionals play a pivotal role in validating DTx solutions and influencing prescribing behaviors, which helps solidify their place as legitimate medical products. However, challenges remain. For example, in Germany, while 70% of doctors are aware of digital health applications (DiGAs), only 7% actively prescribe them. This highlights the need for continuous education and engagement.
"Identifying the most influential KOLs and establishing local partnerships with healthcare providers and systems in different regions, will be important to drive adoption when scaling DTx."
- Anna Mangan, Digital Health Community Manager at HealthXL
Collaborating with local providers during the development phase ensures that DTx solutions integrate seamlessly into existing clinical workflows, a critical factor for long-term adoption.
Public-private partnerships (PPPs) offer another avenue for fostering collaboration. A notable example is the 2023 partnership between Unity Health Toronto and AI company Signal 1. Together, they implemented CHARTWatch, an AI-based patient monitoring tool. Unity Health Toronto took a minor equity stake in the company, serving as a "living lab" to refine the tool’s clinical applications and scale its use. This approach aligned incentives and built capacity for sustainable growth.
AIH LLC: Digital Therapeutics Innovation in Practice
AIH LLC stands out as a leader in digital therapeutics, showcasing how partnerships and cutting-edge technology can create practical solutions for managing chronic diseases.
AI-Powered Wearables for Chronic Disease Management
AIH LLC is transforming the way chronic diseases are managed with its real-time, wearable monitoring devices. Their products include the aiSpine, designed to monitor posture and spine health, and the aiRing, which tracks vital signs. Both devices seamlessly integrate with the AIH Health App, creating a comprehensive ecosystem for remote monitoring.
Instead of relying on subjective self-reporting, these wearables provide accurate metrics like joint angles, symmetry, and endurance. They also offer real-time clinical feedback through audio and visual cues, enabling healthcare providers to monitor patient progress remotely. Built on patented algorithms and backed by international certifications, the technology ensures clinical reliability across various markets. With the global digital therapeutics market projected to hit $11.98 billion by 2026, AIH LLC is addressing the increasing demand for scalable, evidence-based healthcare solutions. This real-time monitoring not only enhances patient care but also fosters stronger collaboration between clinicians and regulatory bodies.
Working with Healthcare Professionals and Regulators
AIH LLC amplifies its impact through strategic partnerships with healthcare professionals and regulators. The company provides physician training programs to help integrate digital therapeutics into existing clinical workflows, easing the adoption process.
Their approach includes offering FDA certification consultation, maintaining strict quality control, and ensuring compliance with market standards. This focus on regulatory alignment is especially relevant, as the U.S. FDA approved 235 new AI-enabled devices in 2024 – a record high. Additionally, AIH LLC operates the Global Medical Bridge, a platform that connects international patients with advanced diagnostic and treatment options in the United States.
Conclusion: The Future of Digital Therapeutics Through Partnerships
The lessons drawn from successful digital therapeutics partnerships highlight a clear path toward scalable, patient-focused healthcare solutions.
Key Takeaways for Stakeholders
Several common threads run through successful digital therapeutics collaborations. When tech companies and pharmaceutical firms combine their strengths, the results can be transformative. Tech companies contribute agility and cutting-edge solutions, while pharmaceutical partners bring clinical expertise, regulatory know-how, and financial backing. Partnerships like these showcase how working together leads to lasting value.
A strong focus on evidence is essential. According to a study on European digital health companies, 86% of digital therapeutics leaders identified demonstrating health impact as the top factor for scaling. This highlights the need to involve Key Opinion Leaders (KOLs) early and to understand the distinct roles of stakeholders – clinicians as validators, patients as users, and payers as choosers – crafting targeted value propositions for each group.
The shift from one-off deals to broader ecosystem partnerships is another important trend. Between 2014 and March 2023, the digital therapeutics market saw over 400 partnership deals, with distribution partnerships making up 25–50% of annual collaborations. Building ecosystems that integrate pharmaceutical companies (for research and funding), commercial distributors, researchers (for evidence generation), and technical service providers offers a more sustainable growth model.
What’s Next for Digital Therapeutics
Looking ahead, the sector must tackle emerging challenges with the same strategic focus. The next phase of digital therapeutics will depend on creating collaboration models that go beyond short-term pilots. With Europe’s aging population – projected to have nearly 30% of its citizens aged 65 or older by 2050 – the demand for chronic disease management solutions will only grow. This demographic trend opens the door for partnerships focused on long-term care, remote monitoring, and personalized treatments.
Regulatory alignment will also play a critical role. Companies can tap into their pharmaceutical partners’ influence to push for clearer reimbursement pathways. A prime example is the $500 million partnership between Boehringer Ingelheim and Click Therapeutics to develop schizophrenia treatments. Such large-scale commitments illustrate how collaboration can help navigate complex regulatory hurdles. Success in this evolving space will require patience, shared goals, and a focus on solving patient problems rather than simply showcasing technology.
FAQs
What makes a DTx partnership worth pursuing?
A digital therapeutics (DTx) partnership makes sense when it supports strategic objectives such as product development, entering new markets, or seamless integration into healthcare systems. These partnerships thrive on combining complementary expertise, securing robust clinical evidence, and navigating complex regulatory requirements effectively.
Collaborating with healthcare stakeholders is a key ingredient for driving innovation and achieving measurable health improvements. A well-structured partnership can speed up clinical validation, streamline regulatory approvals, and boost market adoption. This creates a solid foundation for both therapeutic advancements and long-term commercial success.
How do DTx companies get reimbursement from payers?
Digital therapeutics (DTx) companies face a multi-step process to secure reimbursement, which revolves around three key areas: coding, coverage, and demonstrating value.
Recent changes, such as the 2025 Physician Fee Schedule, have introduced reimbursable codes specifically for prescription digital therapeutics (PDTs). However, despite this progress, coverage for these therapies is still limited, creating challenges for broader adoption.
To succeed, DTx companies must focus on proving their worth. This often means providing solid evidence of clinical outcomes and cost savings. At the same time, navigating complex regulatory pathways is essential. Building strong partnerships with payers and healthcare stakeholders is another critical step, as these relationships help integrate digital therapeutics into existing payment structures.
What are the key data privacy steps for scaling in the U.S.?
To successfully expand digital therapeutics across the U.S., it’s essential to follow federal and state data regulations, establish robust security protocols to protect patient information, and be transparent about how data is managed. Focusing on these areas helps build trust while staying compliant with legal standards.