Machine learning is transforming spine care and chronic disease management. Here’s what you need to know:
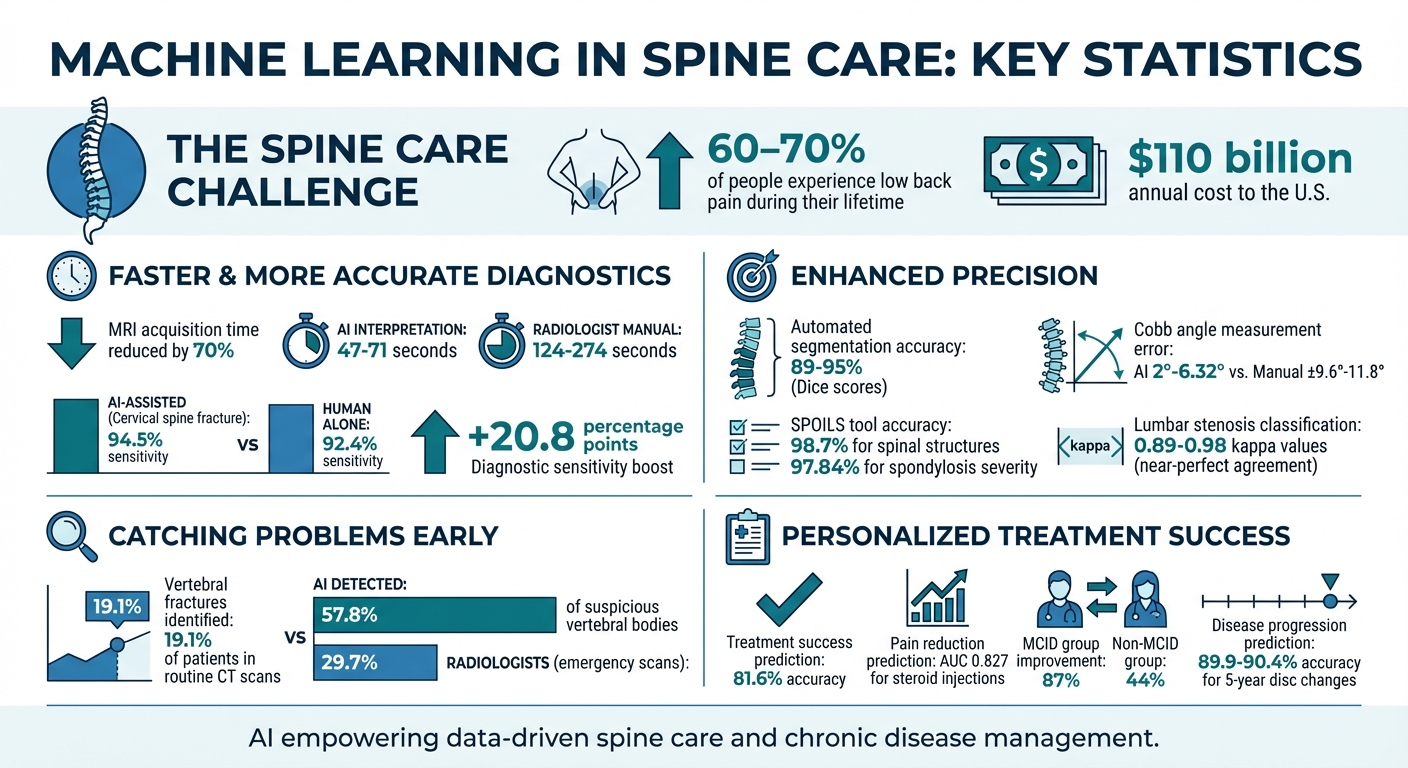
- Low back pain affects 60-70% of people during their lives and costs the U.S. around $110 billion annually.
- AI is helping doctors analyze medical images faster and more accurately, reducing MRI times by 32.3% and boosting diagnostic sensitivity for spinal issues by 20.8 percentage points.
- Algorithms can predict disease progression, personalize treatments, and improve patient outcomes. For example, AI tools achieve 94.5% sensitivity in detecting cervical spine fractures and predict treatment success with 81.6% accuracy.
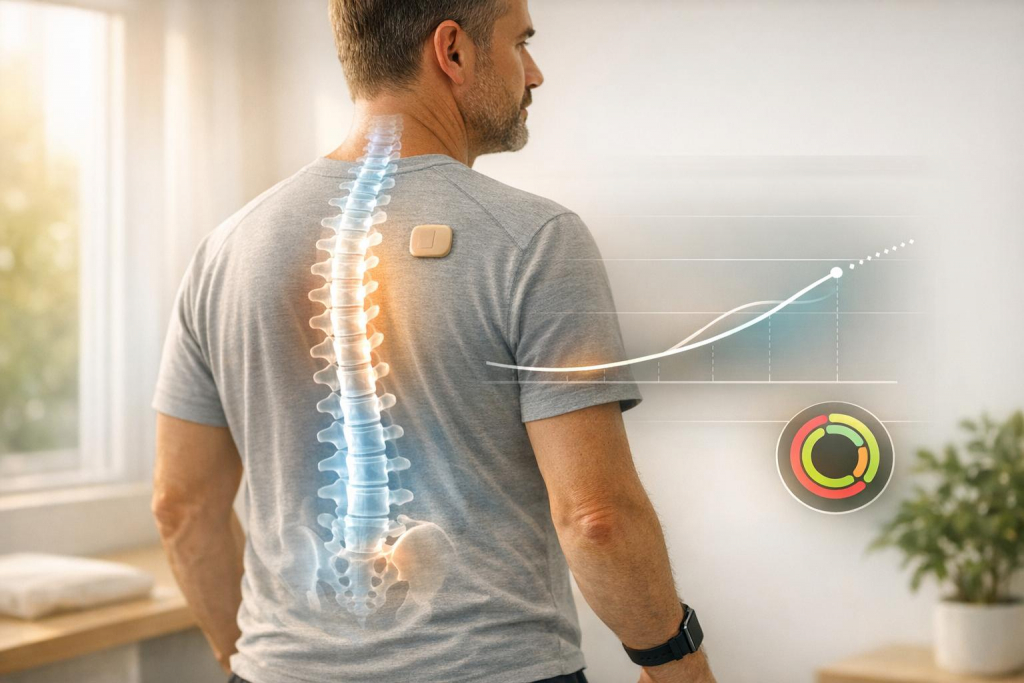
- Wearable devices like aiSpine track posture and recovery in real time, helping tailor rehabilitation.
While challenges like limited datasets, regulatory hurdles, and model transparency remain, AI is empowering healthcare providers with data-driven insights to improve care for spine conditions and chronic diseases.
Machine Learning Impact on Spine Care: Key Statistics and Improvements
Artificial Intelligence and Machine Learning For Spine Surgery
sbb-itb-44aa802
How Machine Learning Improves Spine Diagnostics
Machine learning is transforming the way spine problems are diagnosed, offering faster and more precise image analysis compared to manual methods. These algorithms can process MRIs, CT scans, and X-rays to identify conditions like herniated discs, vertebral fractures, and spinal stenosis. In many cases, they pick up on subtle abnormalities that might be missed by radiologists.
One of the most impressive advancements is the speed. AI systems can interpret lumbar spinal stenosis in just 47 to 71 seconds, while radiologists doing the same task manually take between 124 and 274 seconds. Additionally, deep learning techniques have reduced spine MRI acquisition times by up to 70%, all without sacrificing diagnostic quality. This not only shortens appointments but also lowers costs and makes the process more comfortable for patients. These rapid analyses pave the way for even more advanced automated segmentation techniques, which we’ll explore next.
Automated Analysis of Spinal Imaging
Machine learning shines when it comes to breaking down complicated spinal images into measurable sections. Algorithms can automatically segment vertebrae, intervertebral discs, and the spinal canal with precision levels ranging from 89% to 95%, as measured by Dice scores. This kind of segmentation allows for immediate calculations of critical metrics, like the Cobb angle for scoliosis. The mean error for these automated measurements is only 2° to 6.32°, compared to the ±9.6° to ±11.8° variability seen with manual methods.
A standout example comes from researchers at the All India Institute of Medical Sciences in Raebareli, who developed SPOILS using a DeepLabV3+ segmentation model. This tool achieved 98.7% accuracy in identifying spinal structures and predicted spondylosis severity with 97.84% accuracy.
These tools also act as a "second reader" for radiologists. For instance, in evaluating cervical spine fractures, AI-assisted interpretation reached a 94.5% sensitivity, compared to 92.4% for human assessments alone. By delivering such accurate measurements, machine learning also plays a vital role in detecting spinal abnormalities early, often catching issues that might otherwise go unnoticed.
Early Detection of Spinal Abnormalities
Machine learning isn’t just about measurements – it’s also about spotting problems early. These algorithms excel at identifying subtle irregularities in routine scans, often before they become apparent to the human eye. This capability has proven especially useful for "opportunistic screening", where spinal issues are identified in scans originally taken for other reasons, such as chest or abdominal evaluations.
In 2023, Ong et al. tested a machine learning algorithm on 4,461 patients undergoing routine CT scans. The algorithm identified vertebral fractures in 19.1% of the cohort, while hospital radiologists had only documented 49% of these fractures in their initial reports. Similarly, in emergency whole-body CT scans, AI identified 57.8% of suspicious vertebral bodies, compared to just 29.7% detected by radiologists.
"AI and ML algorithms enhance image analysis by identifying subtle patterns and abnormalities that may be overlooked by the human eye." – MDPI Bioengineering
A great example of this technology is the SpineNet model, developed by Jamaludin et al. This model automates the labeling of vertebral bodies and grades disc degeneration using the Pfirrmann scale in T2-weighted MRI scans. External validation has shown its reliability matches that of expert radiologists in detecting disc narrowing, marrow changes, and foraminal stenosis. For lumbar stenosis classification, AI models have achieved kappa values between 0.89 and 0.98, indicating an almost perfect level of agreement with subspecialized radiologists.
Predicting Disease Risk and Progression
Machine learning is reshaping spine care by predicting which patients are at risk for severe conditions. These advanced models process a wide range of data – like electronic health records, lab results, imaging scans, and patient-reported pain levels – to generate personalized risk scores. The aim? To identify potential problems early, long before they turn into chronic issues or require urgent interventions.
Risk Assessment Using Patient Health Data
Predictive models excel at analyzing data volumes far beyond human capacity. For instance, a Random Forest model used in a multicenter study reached an Area Under the Curve (AUC) of 0.887 in predicting spinal cord injury in patients with cervical spondylosis. Interestingly, the model pinpointed monocyte count as the top predictor, followed by white blood cell count, erythrocyte sedimentation rate, and C-reactive protein levels.
What sets machine learning apart is its ability to uncover complex, non-linear relationships across hundreds of variables – no preset assumptions needed. Algorithms like Random Forest and Deep Neural Networks have even predicted the 5-year progression of disc bulging and Pfirrmann grading with accuracies ranging from 89.9% to 90.4%.
"Machine learning can be used to facilitate personalized medicine via its precise predictions." – ScienceDirect, World Neurosurgery
These technologies also incorporate real-time data from wearable devices. For example, AIH LLC‘s aiSpine and aiRing devices continuously monitor posture and vital signs, feeding this information into the AIH Health App. By combining wearable data with imaging and lab results, clinicians gain a more detailed view of a patient’s condition and can track their trajectory over time. This integrated approach allows healthcare providers to shift from reactive care to proactive, tailored interventions.
Enabling Earlier Medical Interventions
By predicting risks, machine learning enables earlier and more targeted treatments for high-risk patients. Between September 2021 and December 2022, researchers from Cortexx Medical Intelligence and the ELSAN Group tested the SuMO (Surgery Medical Outcomes) platform on 119 patients undergoing lumbar decompression. Using preoperative data like Oswestry Disability Index scores, medication history, and work stress levels, the platform’s deep neural network predicted which patients would achieve a Minimal Clinically Important Difference (MCID) with 81.6% accuracy. Patients who reached the MCID saw their disability scores drop by 87% (from 38 to 5) over 240 days, compared to a 44% drop in those who did not.
"AI-based algorithms may help physicians in their future daily practice by addressing personalized patient care." – Arthur André, Frontiers in Surgery
Deep learning models are also improving treatment decisions for lumbar spine cases. For example, they can predict the success of transforaminal epidural steroid injections with an AUC of 0.827. This helps doctors identify which patients are likely to benefit from injections and which may need alternative treatments. The result? More efficient workflows, fewer unnecessary procedures, and better allocation of resources to patients who need them most. This shift toward proactive care is paving the way for more customized treatment plans.
Personalized Treatment and Rehabilitation Programs
Machine learning is changing the game in healthcare by creating treatment plans tailored to individual patients. Instead of applying a standard approach, these plans now consider factors like genetics, imaging results, clinical history, demographics, and lifestyle habits to deliver more precise care.
Creating Patient-Specific Treatment Plans
Treatment decisions are increasingly guided by predicted success rates rather than generic guidelines. For example, researchers Kim et al. developed a deep learning model that analyzed T2-weighted sagittal lumbar spine MRI data to predict how patients would respond to transforaminal epidural steroid injections for chronic lumbosacral radicular pain. This model achieved an AUC of 0.827, accurately identifying patients likely to experience at least a 50% reduction in pain within two months. This approach not only helps avoid unnecessary procedures for patients unlikely to benefit but also allows for quicker implementation of alternative treatments for those in need.
"Personalized medicine is defined as medical treatment tailored toward the patient’s intrinsic characteristics (e.g., genetics, demographic factors) rather than simply the diagnosis itself." – World Neurosurgery
While earlier methods focused on predicting surgical outcomes, today’s personalized treatment strategies also include psychosocial factors like work stress, family issues, and depression. These variables, which significantly affect long-term recovery, are analyzed alongside physical health data to create well-rounded treatment plans addressing both medical and lifestyle aspects of spine care. Once a plan is in place, continuous monitoring ensures treatments evolve based on the patient’s progress.
Real-Time Monitoring with AI-Powered Wearables
Wearable devices have revolutionized patient monitoring by providing objective, continuous data. Unlike traditional self-reported activity levels, which can vary by as much as 50%, wearables deliver accurate measurements of steps, distance, posture, and movement patterns.
A study at Hayaishi Hospital tested the Fitbit Inspire HR on 71 participants with chronic musculoskeletal pain. The device tracked activity 24/7, and physical therapists reviewed the data weekly, encouraging patients to increase their activity by 10% each week. After six months, the wearable group reported significantly lower pain levels (5.1 vs 6.3 in the control group) and showed improvements on the Pain Catastrophizing Scale and Hospital Anxiety and Depression Scale.
Devices like AIH LLC’s aiSpine and aiRing take this a step further. The aiSpine monitors spinal alignment in real time, sending vibration alerts when poor posture is detected, while the aiRing tracks vital signs during various activities. Both devices feed data into the AIH Health App, where algorithms adjust rehabilitation protocols on the fly. This allows clinicians to make timely interventions when a patient’s recovery veers off course. The result? Adaptive therapy that evolves with the patient’s progress rather than sticking to a fixed schedule.
These advancements in personalized treatment and wearable technology represent a major shift in spine care – from reactive approaches to proactive, data-driven solutions that prioritize the individual needs of each patient.
Implementation Challenges and Future Development
Turning promising lab results into practical clinical tools is far from straightforward. Several key challenges slow the transition, ranging from technical hurdles in model development to navigating the complex regulatory landscape.
Data Requirements and Model Training
Machine learning models thrive on large and diverse datasets, yet spine research often relies on small, narrowly focused datasets. This limitation affects both the accuracy and the ability to generalize findings across varied patient populations. To make matters more complicated, supervised learning methods require labeled data – labels that only medical experts can provide. This process is often time-intensive, costly, and prone to human error. Additionally, the wide variability in patient conditions further complicates model accuracy.
Another major issue is validation. Most models are validated internally, meaning they’re tested on the same dataset used for training. While this is a step forward, it falls short of proving the model’s reliability in real-world scenarios. External validation – testing models on new, independent datasets – is rare, largely due to challenges in sharing data between institutions. However, external validation is critical before any model can be recommended for clinical use.
"Models should not be recommended for clinical use before external validity is established." – Fabio Galbusera, Spine Center, Schulthess Clinic
The "black box" nature of deep learning models also creates distrust among clinicians. These systems often lack transparency in how they arrive at decisions, making it hard for healthcare professionals to fully rely on them. Explainable AI (XAI) frameworks, like SHAP, aim to make these processes more transparent, which is crucial for regulatory acceptance. However, their adoption in clinical settings remains limited. To add to the complexity, subjective outcome measures such as pain or functional status – often influenced by psychological and socioeconomic factors – are difficult for AI to assess without bias.
These challenges in data and model training directly affect the rigorous standards required for regulatory approval.
Regulatory Approval and Clinical Validation
Even as AI shows promise in diagnostics, regulatory frameworks must adapt to validate these tools across diverse clinical environments. For instance, the EU AI Act classifies most diagnostic and therapeutic AI systems as "high-risk", requiring stringent third-party assessments and CE marking before they can enter the market. Similarly, in the United States, developers must navigate the FDA’s demanding approval process, which involves extensive documentation on system architecture, training methods, and validation outcomes.
As of October 2023, the FDA had approved 691 AI/ML-enabled medical devices. A noteworthy example is Medtronic‘s UNiD Spine Analyzer, the first FDA-approved AI tool for spine surgery. Approved in July 2022, it uses algorithms trained on 10,000 surgical cases to assist surgeons with preoperative planning and predicting outcomes. Another example is CoLumbo, which earned CE marking in the EU in 2021 for detecting conditions like disc herniation from MRI scans. To later secure FDA approval via the 510(k) pathway, developers used separate training datasets from the EU and the US to reduce regional bias.
| Challenge Category | Specific Technical Barrier | Impact on Clinical Practice |
|---|---|---|
| Data | Small sample sizes and lack of diversity | Reduced ability to generalize across populations |
| Validation | Limited external testing | Uncertainty about real-world safety and effectiveness |
| Interpretability | Opaque decision-making in deep learning | Low clinician trust and regulatory hurdles |
| Deployment | Real-time processing demands in the OR | Requires advanced hardware and workflow adjustments |
| Ethics | Bias from unrepresentative datasets | Risk of unequal treatment outcomes for marginalized groups |
The FDA has introduced Predetermined Change Control Plans (PCPCs) to address the unique challenges posed by adaptive algorithms. Meanwhile, South Korea’s Ministry of Food and Drug Safety (MFDS) recently streamlined its approval process, allowing lower-risk medical software to bypass clinical trials, making it easier for AI-based diagnostic tools to reach the market.
Looking ahead, future developments aim to integrate multimodal data, blending imaging, clinical history, genomics, and real-time wearable data to enable more personalized spine care. Generative AI models are also being explored to create synthetic datasets, which could help alleviate the scarcity of high-quality spine research data. Enhanced explainability and better-aligned regulatory frameworks will play a key role in unlocking the full potential of AI in treating spine conditions and other chronic diseases.
Conclusion
Machine learning is transforming spine care in profound ways, from diagnosis to treatment and recovery. Its ability to detect conditions like vertebral fractures and disc herniations with accuracy on par with, or even better than, radiologists is a game-changer. Beyond precision, it slashes the time needed for manual analysis, paving the way for quicker and earlier interventions. In many cases, degenerative changes can now be identified before symptoms even appear, giving patients a head start on treatment.
But it doesn’t stop at diagnosis. Machine learning enables personalized care by analyzing individual factors – like genetics, demographics, and medical history – to predict the most effective treatments for each patient. This tailored approach is delivering real results: patients in the MCID group showed an 87% improvement rate compared to 44% in others. It’s a shift away from one-size-fits-all guidelines toward care that’s truly individualized.
The economic benefits are just as compelling. By enhancing surgical planning and reducing complications, machine learning offers a way to manage rising healthcare costs. Tools like wearable devices and remote monitoring systems also play a crucial role, allowing for continuous patient tracking and adaptive rehabilitation. These innovations not only boost adherence but also support better long-term outcomes.
While challenges like data quality and regulatory issues remain, machine learning is proving to be an essential tool in modern spine care. It’s not about replacing healthcare professionals but empowering them with data-driven insights to deliver more effective and personalized care for patients dealing with spine conditions and chronic diseases.
FAQs
Will AI replace my radiologist or spine doctor?
AI is playing a growing role in spinal imaging and care, offering tools to improve diagnostics and assist in decision-making. That said, it’s important to note that AI is meant to support specialists like radiologists and spine doctors – not replace them. While it helps boost efficiency, predict risks, and contribute to personalized treatment plans, AI lacks the nuanced clinical judgment that comes from years of medical training and experience. Instead, it works alongside healthcare professionals, enhancing their expertise to improve patient care and optimize workflows.
How accurate is machine learning for spotting spine problems early?
Machine learning is making waves in the early detection of spine issues. Studies reveal that it can accurately predict spinal cord injuries and assist in diagnoses, paving the way for quicker interventions and customized treatments. This progress is reshaping spine care by enhancing patient outcomes and creating more tailored approaches to treatment.
How do wearables like aiSpine and aiRing help with recovery at home?
Wearable devices such as aiSpine and aiRing are transforming home recovery by offering round-the-clock health monitoring and tailored feedback. The aiSpine focuses on tracking posture and spinal alignment, which can help reduce the risk of further injuries. Meanwhile, the aiRing keeps tabs on vital signs like heart rate and temperature.
Both devices sync seamlessly with the AIH Health App, delivering real-time data to both patients and healthcare providers. This setup allows for personalized care, early detection of potential issues, and a smoother recovery process – all without the need for constant in-person appointments.